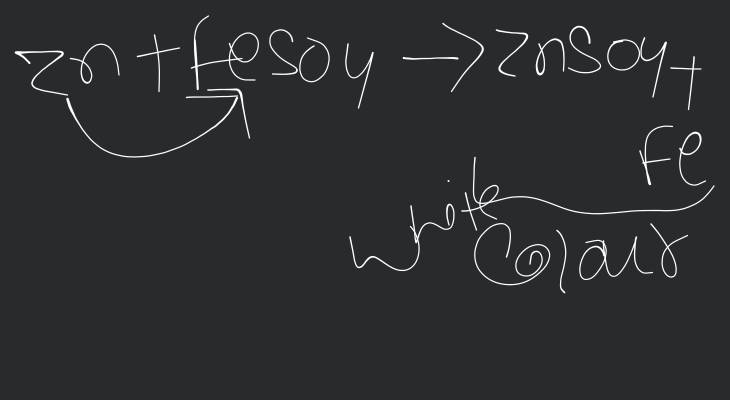Question
Easy
Solving time: 2 mins
What would you observe when Zinc is added to a solution of Iron(II) sulphate? Write chemical reaction that takes place.
Found 2 tutors discussing this question
Discuss this question LIVE
12 mins ago
 Text solution
Text solution Verified
Verified
When zinc is added to solution of Iron (II) sulphate, the colour of iron sulphate solution changes. It is because zinc is more reactive than iron, therefore it displaces iron from it's solution of iron sulphate and a grey precipitate of iron and a colourless solution of zinc sulphate is formed.
Was this solution helpful?
79
Share
Report

One destination to cover all your homework and assignment needs
Learn Practice Revision Succeed

Instant 1:1 help, 24x7
60, 000+ Expert tutors

Textbook solutions
Big idea maths, McGraw-Hill Education etc

Essay review
Get expert feedback on your essay

Schedule classes
High dosage tutoring from Dedicated 3 experts
Practice more questions from Science (NCERT)
Q1
Food cans are coated with tin and not with zinc because:
Q2
What would you observe when Zinc is added to a solution of Iron(II) sulphate? Write chemical reaction that takes place.
Q3
You are given a hammer, a battery, a bulb, wires and a switch.
(a) How could you use them to distinguish between samples of metals and non-metals?
(b) Assess the usefulness of these tests in distinguishing between metals and non-metals.
View all(a) How could you use them to distinguish between samples of metals and non-metals?
(b) Assess the usefulness of these tests in distinguishing between metals and non-metals.
Practice questions from Science (NCERT)
Question 1
Medium
Views: 6,039
(i) is a liquid at room temperature
(ii) can be easily cut with a knife.
(iii) is the best conductor of heat.
(iv) is a poor conductor of heat.
Question 2
Easy
Views: 5,984
Question 3
Easy
Views: 5,734
Practice more questions from Metals and Non-metals
Question 1
Easy
Views: 5,709
(a) Name an alloy of copper. State its chemical composition and any one use.
(b) Explain why, when a copper object remains in damp air for a considerable time, a green coating is formed on its surface. What is this process known as?
Question 2
Easy
Views: 6,024
Explain giving one example, how highly reactive Metals and Non-metals (which are high up in the reactivity series) are extracted.
Question 4
Easy
Views: 6,001
Two non-Metals and Non-metals combine with each other by the sharing of electrons to form a compound X.
(a) What type of chemical bond is present in X?
(b) State whether X will have a high melting point or low melting point.
(c) Will it be a good conductor of electricity or not?
(d) Will it dissolve in an organic solvent or not?
Practice questions on similar concepts asked by Filo students
Question 1
Views: 5,312
Question 2
Views: 6,013


Stuck on the question or explanation?
Connect with our Science tutors online and get step by step solution of this question.
231 students are taking LIVE classes
| Question Text | What would you observe when Zinc is added to a solution of Iron(II) sulphate? Write chemical reaction that takes place. |
| Updated On | Mar 19, 2023 |
| Topic | Metals and Non-metals |
| Subject | Science |
| Class | Class 10 |
| Answer Type | Text solution:1 Video solution: 1 |
| Upvotes | 227 |
| Avg. Video Duration | 2 min |