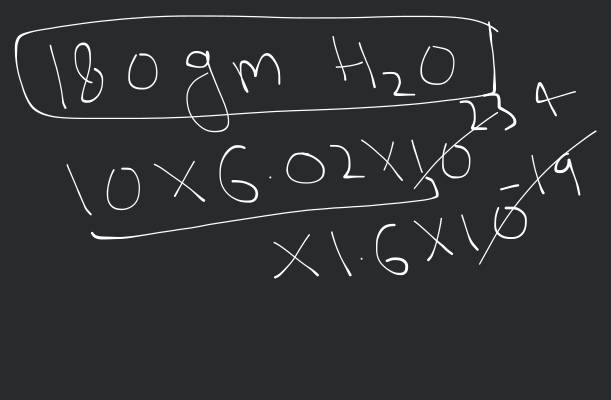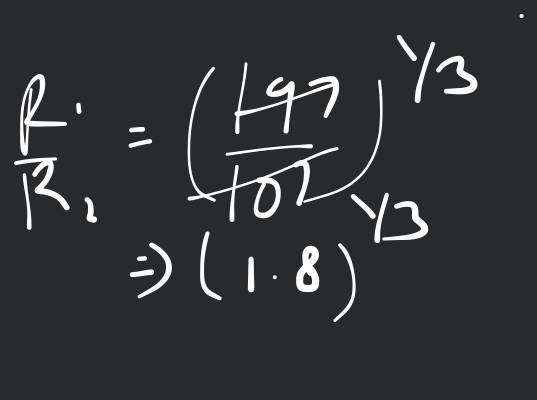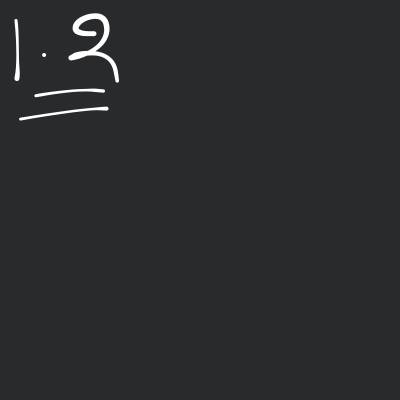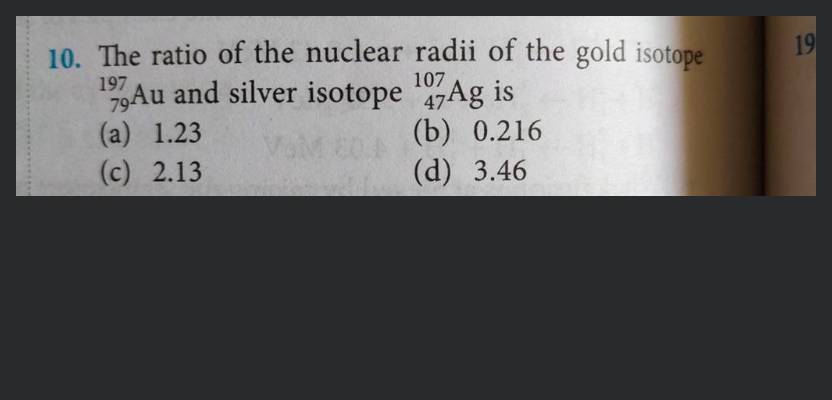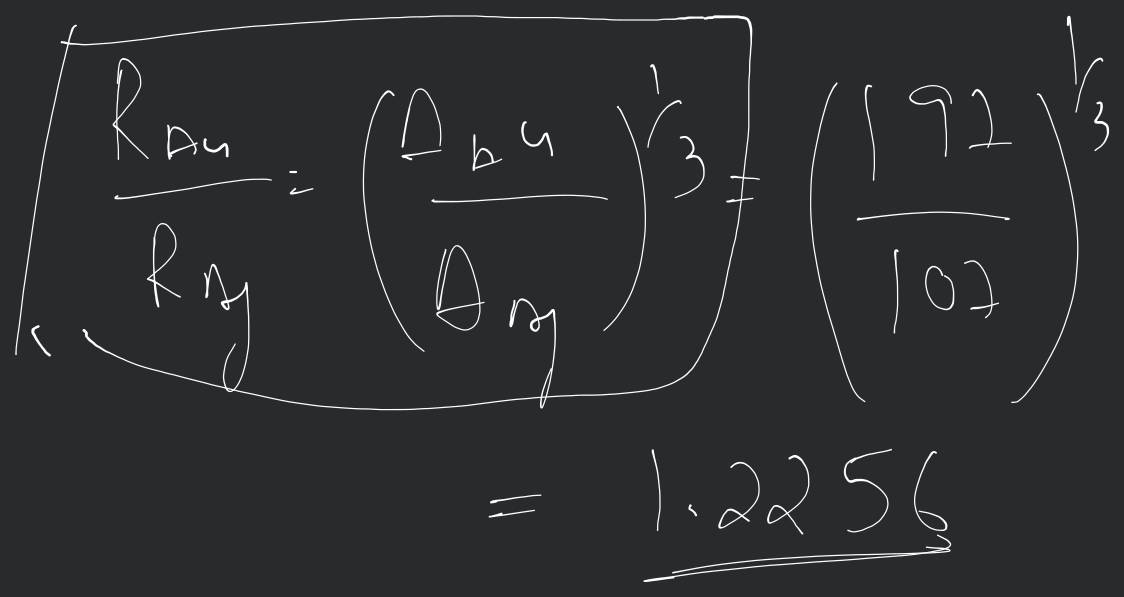Question
Easy
Solving time: 3 mins
Obtain approximately the ratio of the nuclear radii of the gold isotope and the silver isotope .
Found 6 tutors discussing this question
Discuss this question LIVE
14 mins ago
 Text solution
Text solution Verified
Verified
Given: The mass number of the gold isotope is .
The mass number of tehe silver isotope is .
The mass number of tehe silver isotope is .
The ratio of nuclear radii is given as:
Was this solution helpful?
51
Share
Report
Filo tutor solutions (4)
Learn from their 1-to-1 discussion with Filo tutors.
22 mins
Uploaded on: 2/14/2023
Was this solution helpful?
105
Share
Report
5 mins
Uploaded on: 3/9/2023
Was this solution helpful?
58
Share
Report

One destination to cover all your homework and assignment needs
Learn Practice Revision Succeed

Instant 1:1 help, 24x7
60, 000+ Expert tutors

Textbook solutions
Big idea maths, McGraw-Hill Education etc

Essay review
Get expert feedback on your essay

Schedule classes
High dosage tutoring from Dedicated 3 experts
Practice more questions from Physics Part-II (NCERT)
Q1
The half-life of is 28 years. What is the disintegration rate of 15 mg of this isotope?
Q2
Obtain approximately the ratio of the nuclear radii of the gold isotope and the silver isotope .
Q3
Find the Q-value and the kinetic energy of the emitted - Particle in the -decay of (a) and (b) .
Given:
,
,
,
,
Practice questions from Physics Part-II (NCERT)
Question 1
Medium
Views: 6,372
(a)
(b)
Question 2
Easy
Views: 5,479
.
Question 3
Medium
Views: 6,140
(a) Calculate the energy released in MeV in this reaction from the data:
(b) Consider the radius of both deuterium and tritium to be approximately 2.0 fm. What is the kinetic energy needed to overcome the coulomb repulsion between the two nuclei? To what temperature must the gas be heated to initiate the reaction? (Hint: Kinetic energy required for one fusion event =average thermal kinetic energy available with the interacting particles = 2(3kT/2); k = Boltzmans constant, T = absolute temperature.)
Question 4
Medium
Views: 5,950
(a) Two stable isotopes of lithium and have respective abundances of 7.5% and 92.5%. These isotopes have masses 6.01512 u and 7.01600 u, respectively. Find the atomic mass of lithium.
(b) Boron has two stable isotopes, and . Their respective masses are 10.01294 u and 11.00931 u, and the atomic mass of boron is 10.811 u. Find the abundances of and B.
Practice more questions from Nuclei
Question 2
Medium
Views: 5,687
Practice questions on similar concepts asked by Filo students
Question 1
Views: 5,296
Question 2
Views: 5,155
Question 3
Views: 5,590


Stuck on the question or explanation?
Connect with our Physics tutors online and get step by step solution of this question.
231 students are taking LIVE classes
| Question Text | Obtain approximately the ratio of the nuclear radii of the gold isotope and the silver isotope . |
| Updated On | Apr 21, 2023 |
| Topic | Nuclei |
| Subject | Physics |
| Class | Class 12 |
| Answer Type | Text solution:1 Video solution: 4 |
| Upvotes | 413 |
| Avg. Video Duration | 8 min |