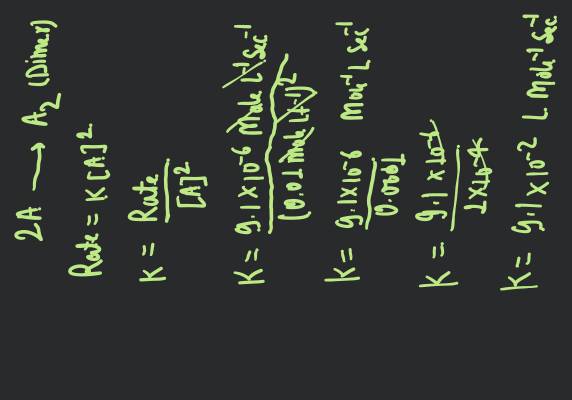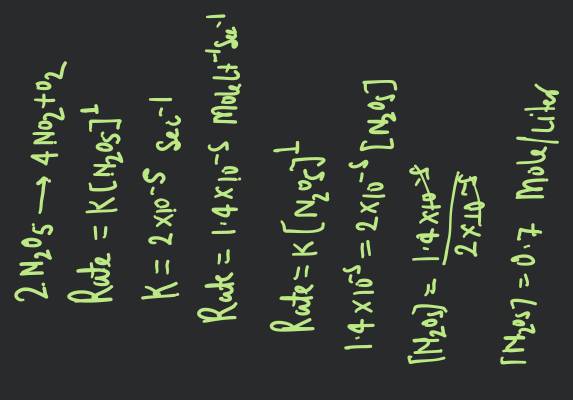Question
Question asked by Filo student

The order of reaction is decided by
- temperature
- mechanism of reaction as well as relative concentration of reactants
- molecularity
- pressure.
Found 5 tutors discussing this question
Discuss this question LIVE
8 mins ago
Filo tutor solutions (3)
Learn from their 1-to-1 discussion with Filo tutors.
2 mins
Uploaded on: 3/20/2023
Was this solution helpful?
55
Share
Report
12 mins
Uploaded on: 4/16/2023
Was this solution helpful?
55
Share
Report

One destination to cover all your homework and assignment needs
Learn Practice Revision Succeed

Instant 1:1 help, 24x7
60, 000+ Expert tutors

Textbook solutions
Big idea maths, McGraw-Hill Education etc

Essay review
Get expert feedback on your essay

Schedule classes
High dosage tutoring from Dedicated 3 experts
Practice more questions on Chemical Kinetics
Question 1
Medium
Views: 6,037
One of its examples is the use of ethylene glycol and water mixture as an anti-freezing liquid in the radiator of automobiles.
A solution is prepared by mixing ethanol and water. The mole fraction of ethanol in the mixture is 0.9.
Given, the freezing point depression constant of water
Freezing point depression constant of ethanol
Boiling point elevation constant of water
Boiling point elevation constant of ethanol
Standard freezing point of water
Standard freezing point of ethanol
Standard boiling point of water
Standard boiling point of ethanol
Vapour pressure of pure water
Vapour pressure of pure ethanol
Molecular weight of water
Molecular weight of ethanol
In answering the following questions, consider the solutions to be ideal dilute solutions and solutes to be non-volatile and non-dissociative.
Question 2
Easy
Views: 5,305
Question 3
Medium
Views: 5,195
Match of the rebox pair in Column I with the values given in Column II and select the correct answer using the code given below the lists.
Students who ask this question also asked
Question 1
Views: 5,545
Question 2
Views: 5,771
.
An increase in temperature shows: a. more dissociation of and a decreases in b. less dissociation of and an increases in c. more dissociation of and an increase in d. less dissociation of and a decrease in
Question 4
Views: 5,538


Stuck on the question or explanation?
Connect with our Chemistry tutors online and get step by step solution of this question.
231 students are taking LIVE classes
| Question Text | The order of reaction is decided by |
| Updated On | Jun 1, 2023 |
| Topic | Chemical Kinetics |
| Subject | Chemistry |
| Class | Class 12 |
| Answer Type | Video solution: 3 |
| Upvotes | 240 |
| Avg. Video Duration | 10 min |