Question
Medium
Solving time: 3 mins
Effective nuclear charge is the net attractive force on electrons under Consideration and is equal to: (nuclear charge screening constant). or is calculated by Slater's formula, as given. If one electron is present in the outermost orbit, there will be no screening in that orbital. Each electron contribute, (total electrons minus one electron) present in the outermost shell. A contribution of for each electron is taken in the (n-1)th shell. For all other electrons contribution is 1 for each electron. The screening constant for electron of ( will be :
Found 6 tutors discussing this question
Discuss this question LIVE
14 mins ago
 Text solution
Text solution Verified
Verified
(d) 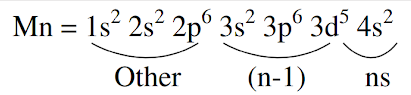
Was this solution helpful?
71
Share
Report

One destination to cover all your homework and assignment needs
Learn Practice Revision Succeed

Instant 1:1 help, 24x7
60, 000+ Expert tutors

Textbook solutions
Big idea maths, McGraw-Hill Education etc

Essay review
Get expert feedback on your essay

Schedule classes
High dosage tutoring from Dedicated 3 experts
Practice questions from Inorganic Chemistry for IIT JEE Main and Advanced (McGraw Hill)
Question 4
Following statements regarding the periodic trends of chemical reactivity of the alkali metals and the halogens are given. Which of these statements gives the correct picture?Medium
Practice more questions from Classification of Elements and Periodicity in Properties
Practice questions on similar concepts asked by Filo students
Question 1
Use the following equations to answer the next four questions:i. ii. iii. iv. v. (a) Which equation describes a physical change?(b) Which equation identifies the reactants and products of a combustion reaction?(c) Which equation is not balanced?(d) Which is a net ionic equation?Question 3
the expression for the energy change. (in joules) such that where and . The spectral lines correspond to Paschen series to
(a) and
(b) and
(c) and Question 4
decrease if the amount o1 diry reactant is taken in excess
ATOMIC, MOLECULAR AND EOUIVALENT MASSES


Stuck on the question or explanation?
Connect with our Chemistry tutors online and get step by step solution of this question.
231 students are taking LIVE classes
| Question Text | Effective nuclear charge is the net attractive force on electrons under Consideration and is equal to: (nuclear charge screening constant). or is calculated by Slater's formula, as given. If one electron is present in the outermost orbit, there will be no screening in that orbital. Each electron contribute, (total electrons minus one electron) present in the outermost shell. A contribution of for each electron is taken in the (n-1)th shell. For all other electrons contribution is 1 for each electron. The screening constant for electron of ( will be : |
| Topic | Classification of Elements and Periodicity in Properties |
| Subject | Chemistry |
| Class | Class 11 |
| Answer Type | Text solution:1 |
| Upvotes | 71 |



