Question
Medium
Solving time: 3 mins

Consider the given reaction the product A is:
Found 6 tutors discussing this question
Discuss this question LIVE
7 mins ago
 Text solution
Text solution Verified
Verified
Halogenation of 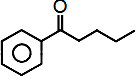 by electrophilic substitution reaction mechanism in presence of Lewis
by electrophilic substitution reaction mechanism in presence of Lewis
acid take place at meta position.
acid take place at meta position.
Was this solution helpful?
43
Share
Report

One destination to cover all your homework and assignment needs
Learn Practice Revision Succeed

Instant 1:1 help, 24x7
60, 000+ Expert tutors

Textbook solutions
Big idea maths, McGraw-Hill Education etc

Essay review
Get expert feedback on your essay

Schedule classes
High dosage tutoring from Dedicated 3 experts
Questions from JEE Mains 2021 - PYQs
Question 2
Easy
Views: 5,590
Question 3
Medium
Views: 5,292
Statements I : Sphalerite is a sulphide ore of zinc and copper glance is a sulphide ore of copper.
Statements II: It is possible to separate two sulphide ores by adjusting proportion of oil to water or by using 'depressants' in a froth flotation method.
Choose the most appropriate answer from the options given below:
Question 4
Easy
Views: 5,661
Statement I: The identification of is carried out by Dimethylglyoxime in the presence of
Statement II: The Dimethylglyoxime is a bidentate neutral ligand.
In the light of the above statements, choose the correct answer from the options given below:
Practice questions from Aldehydes, Ketones and Carboxylic Acids in the same exam
Question 1
Medium
Views: 5,472
The compound which is not formed as a product in the reaction is a:
Practice more questions from Aldehydes, Ketones and Carboxylic Acids
View morePractice questions on similar concepts asked by Filo students
Question 1
Views: 5,221


Stuck on the question or explanation?
Connect with our Chemistry tutors online and get step by step solution of this question.
231 students are taking LIVE classes
| Question Text | Consider the given reaction the product A is: |
| Topic | Aldehydes, Ketones and Carboxylic Acids |
| Subject | Chemistry |
| Class | Class 12 |
| Answer Type | Text solution:1 |
| Upvotes | 43 |




